Indonesia Gearing Up to Produce Homegrown Covid-19 Vaccines
JAKARTA, KOMPAS.com - Indonesia is in the process of producing its own Covid-19 vaccine as part of the country's responses to pandemic amid the vaccine shortage globally.
Two homegrown candidate vaccines are currently being developed by Indonesian scientists. The vaccines comprise the Nusantara vaccine and the Merah Putih (red and white) vaccine. These two candidate vaccines are initiated by former health minister Terawan Agus Putranto and the Research and Technology Ministry/National Research and Innovation Agency (BRIN), respectively.
However, the Nusantara vaccine was already facing controversy at the beginning of its journey. This began when the country's Food and Drug Agency (BPOM) decided to stop the tests of the vaccine at the second and third clinical stages in March following its low efficacy rate and failure to fulfill other requirements in clinical practice.
In efforts to resolve the matter, a memorandum of understanding (MoU) was signed by relevant parties comprising the Ministry of Health, BPOM, and the Indonesian Armed Forces (TNI AD) on Monday, April 19.
The MoU is expected to achieve a better understanding of the purpose of the Nusantara vaccine development. This includes that the development of a dendritic cell-based Covid-19 vaccine will only be used for research and service purposes and cannot be commercialized as it has yet received a permit for distribution from BPOM.

Meanwhile, the Merah Putih vaccine by the Eijkman Institute for Molecular Biology (LMB) and Bio Farma Ltd., which is made with a recombinant protein platform, is expected to be produced in early 2022.
Also read: BPOM: Indonesias Merah Putih Vaccine to Kick Off Production in 2022
"We hope EUA or Emergency Use Authorization [to the Eijkman and Bio Farma Merah Putih Vaccine] will be issued by the first semester of 2022 after the completion of clinical trials. Once that happens, we can start producing the vaccine in the second semester of 2022," BPOM Head Penny Lukito said on Friday, April 16.
The Merah Putih vaccine, named after the country's national flag, does not refer to just one type of vaccine. Instead, the vaccine is developed by a research consortium under the Ministry of Research and Technology/National Research and Innovation Agency (BRIN).
With the development of homegrown vaccines, it is hoped that Indonesia will have sufficient doses to inoculate its citizens in the efforts to contain the spread of Covid-19.
 President Joko Widodo welcomed the arrival of the Red and White Vaccine team at the Bogor Presidential Palace, West Java, September 9, 2020.
President Joko Widodo welcomed the arrival of the Red and White Vaccine team at the Bogor Presidential Palace, West Java, September 9, 2020.
 BPOM Head Penny Lukito attended a working meeting with DPR Commission IX at the Parliament Complex, Senayan, Jakarta, Wednesday, March 10. The meeting discussed government support for the development of the Merah Putih (red and white) vaccine and the Nusantara vaccine.
BPOM Head Penny Lukito attended a working meeting with DPR Commission IX at the Parliament Complex, Senayan, Jakarta, Wednesday, March 10. The meeting discussed government support for the development of the Merah Putih (red and white) vaccine and the Nusantara vaccine. 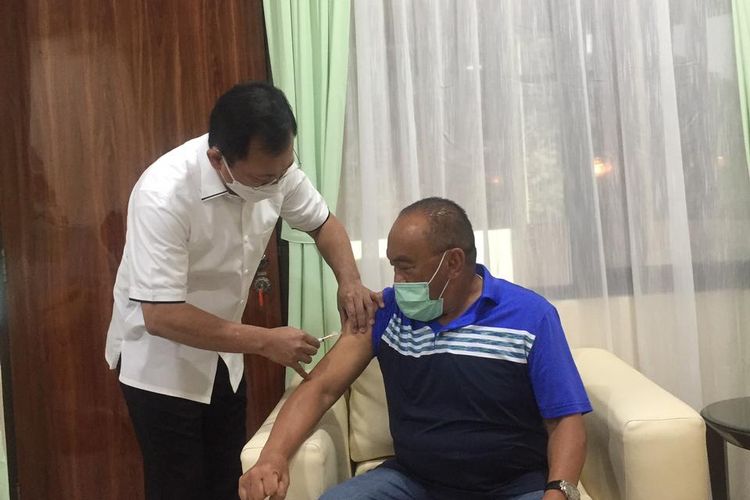 Former Health Minister Terawan Agus Putranto injected the Nusantara vaccine at senior politician Aburizal Bakrie at the Gatot Soebroto Army Hospital, Jakarta, Friday, April 16.
Former Health Minister Terawan Agus Putranto injected the Nusantara vaccine at senior politician Aburizal Bakrie at the Gatot Soebroto Army Hospital, Jakarta, Friday, April 16. Health Minister Budi Gunadi Sadikin, Army Chief of Staff Gen. Andika Perkasa and BPOM Head Penny Lukito signed a Memorandum of Understanding (MoU) related to development of the homegrown Nusantara vaccine, Monday, April 19.
Health Minister Budi Gunadi Sadikin, Army Chief of Staff Gen. Andika Perkasa and BPOM Head Penny Lukito signed a Memorandum of Understanding (MoU) related to development of the homegrown Nusantara vaccine, Monday, April 19.(Writer: Haryanti Puspa Sari, Nicholas Ryan Aditya, Wisnu Nugroho | Editor: Icha Rastika, Krisiandi, Amir Sodikin)
https://www.kompas.com/tren/read/2021/04/20/095144965/akhir-polemik-vaksin-nusantara
Simak breaking news dan berita pilihan kami langsung di ponselmu. Pilih saluran andalanmu akses berita Kompas.com WhatsApp Channel : https://www.whatsapp.com/channel/0029VaFPbedBPzjZrk13HO3D. Pastikan kamu sudah install aplikasi WhatsApp ya.