Baca berita tanpa iklan. Gabung Kompas.com+
Indonesia Receives 1.2 Million Doses of Covid-19 Vaccine from China’s Sinovac

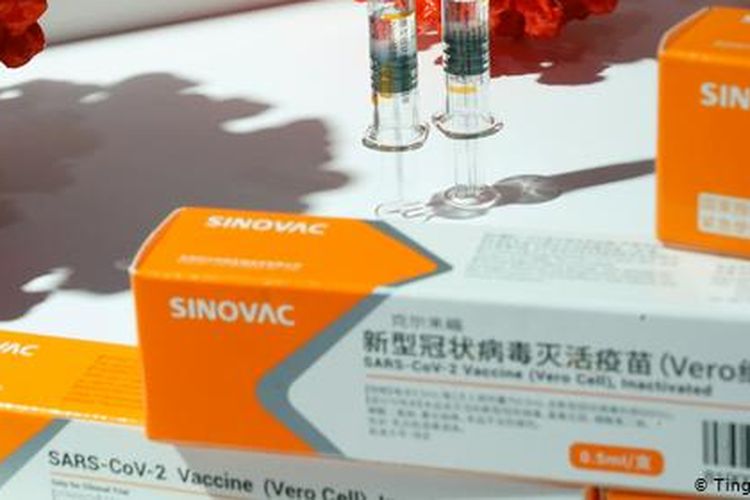
The first shipment of Covid-19 vaccine from Chinese drug maker Sinovac has arrived in Soekarno-Hatta International Airport near Jakarta on Sunday, December 6.
Writer Dezy Rosalia Piri
| Editor Melecio Luib
BPOM head Penny Lukito said that the clinical trial in Bandung went smoothly.
Penny also assured the public that the vaccine has met the halal standards following a recent examination jointly conducted by BPOM, Bio Farma, and the Indonesian Ulema Council (MUI).
Indonesia has also committed to procuring vaccines with other vaccine makers such as Sinopharm in collaboration with G42 in the United Arab Emirates, CanSino, and AstraZeneca.
(Writer: Rakhmat Nur Hakim | Editors: Kristian Erdianto, Icha Rastika)
Pilih idol K-Pop/aktor K-Drama favoritmu & dapatkan Samsung Galaxy Fit3!
Kompas.id

There are no comments
Comment wisely and responsibly. Comments are entirely the responsibility of the commentator as regulated in the ITE Law
Thank You! We have received your report. We will remove comments that conflict with the Community Guidelines and the ITE Law.
More Headlines
Business
August 25, 2023, 07.58 PM
Taiwan's Semiconductor Industry: A Rising Powerhouse with Global Impact
News
April 14, 2023, 11.48 AM
3 Malaysian Nationals Missing after Boat Capsizes Near Indonesia’s Side of Borneo
News
April 13, 2023, 01.56 PM
Indonesia to Deploy Warships during ASEAN Summit in East Nusa Tenggara
News
April 12, 2023, 02.32 PM
Jakarta High Court Rejects Appeal of Ex-Police General Ferdy Sambo Facing Death Sentence
News
April 11, 2023, 05.26 PM
Indonesia Religious School Principal Named Suspect in 14 Students' Sexual Abuse
Culture
April 8, 2023, 05.52 PM
Centuries-old Indonesian Easter Tradition Returns after Pandemic Hiatus
News
April 8, 2023, 05.35 PM
Access to ASEAN Air Transportation Drives Bali's Tourism, Says Statistics Indonesia
Baca berita tanpa iklan. Gabung Kompas.com+
Baca berita tanpa iklan. Gabung Kompas.com+
IN CASE YOU MISSED
Baca berita tanpa iklan. Gabung Kompas.com+
Baca berita tanpa iklan. Gabung Kompas.com+