Indonesia Highlights: Sinopharm Covid-19 Vaccines Arrives in Indonesia | Indonesian Police: Thousands Affected by Used Rapid Test Samples in North Sumatra Indonesia | Indonesian Officials in Bali Depo
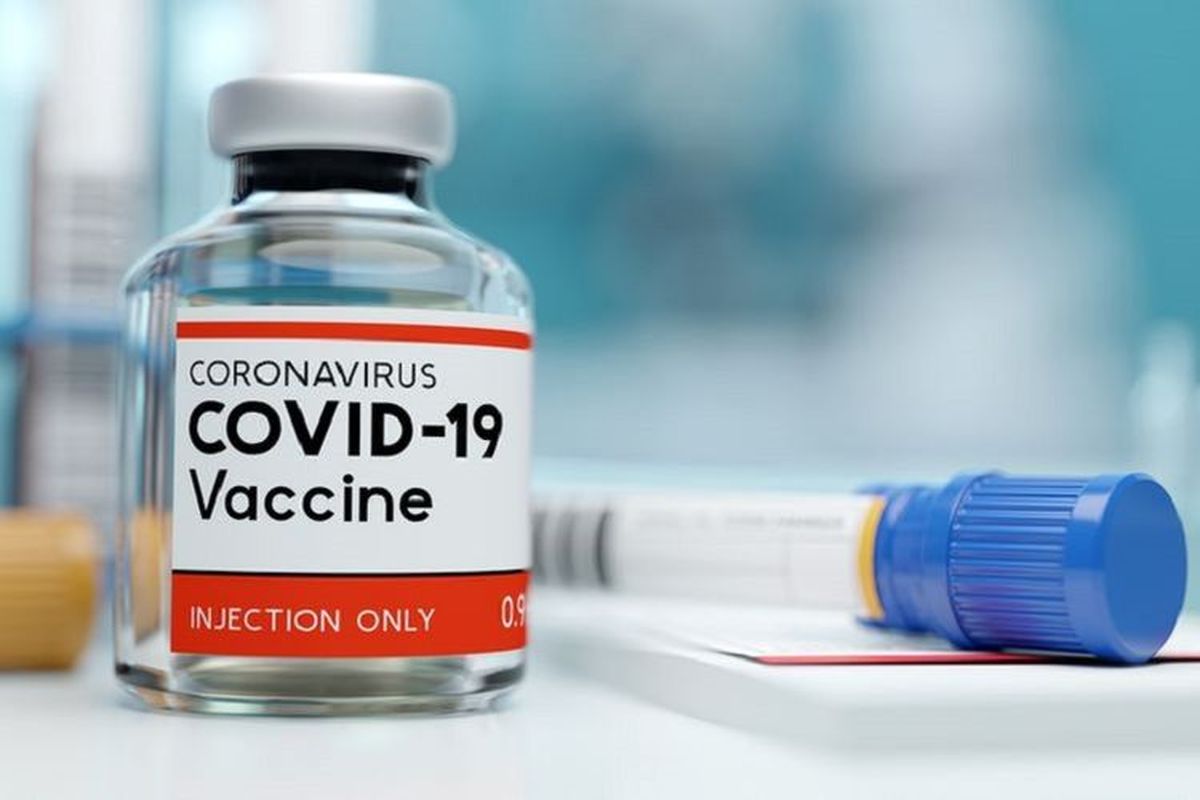
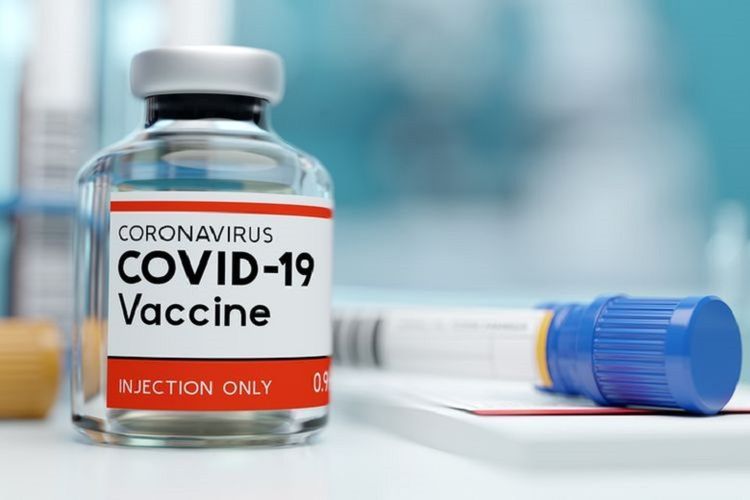
JAKARTA, KOMPAS.com – Indonesia has taken delivery of the Sinopharm vaccine from China, as the country continues to stockpile vaccines to for use in its quest to gain herd immunity against Covid-19.
Indonesian police in North Sumatra continued to find more details of a racket to use old Covid-19 rapid test kits on travelers in the province’s Kualanamu International Airport.
And immigration officials in Bali deport two foreign citizens who tried to get around health protocols with a misguided attempt to find the funny side in the stunt. This and more on Indonesia Highlights:
 BPOM head Penny Lukito announcing the EUA for the Sinopharm vaccine at a press conference on Friday, (30/4/2021)
BPOM head Penny Lukito announcing the EUA for the Sinopharm vaccine at a press conference on Friday, (30/4/2021) Sinopharm Covid-19 Vaccines Arrives in Indonesia
Indonesia is set to increase its stockpile of Covid-19 vaccines, as the country received the first doses of the Sinopharm coronavirus vaccine.
Minister of Communications and Informatics Johnny G. Plate are among the officials who received the shipment, which arrived in Soekarno-Hatta International Airport on Friday, April 30. “
The cargo included 482,400 ready-to use vaccine doses of the Sinopharm China National Pharmaceutical Corporation, and six million bulk vaccines from Sinovac,” said the Nasdem Party [National Democrat] politician.
“This latest consignment of vaccines has brought the number of vaccine doses in Indonesia to 65,500,000 bulk vaccines from Sinovac, as well as 8,448,000 finished product vaccines from Sinovac, Sinopharm and AstraZeneca from our cooperation with the Gavi Covax Facility.”
Indonesia’s Food and Drug Agency or BPOM has issued emergency use authorization [EUA] for Sinopharm’s Covid-19 vaccine on the day of their arrival.
BPOM chief Penny Lukito said the agency’s decision to issue the EUA to the Sinopharm vaccine was based on reports of its positive results in the United Arab Emirates.
“Phase 3 clinical studies carried out on 42 thousand volunteers in the United Arab Emirates has shown that the [Sinopharm] vaccine has an efficacy rate of 78 percent,” she said in a virtual press conference on Friday, April 30.
Penny admitted that the vaccine has some side effects, among them bruises, swelling, headaches, diarrhea, aches and coughs, among other physical effects. But she maintained that they are still mild and only affected 0.01 percent of those who received the vaccine.
Indonesia will use the Sinopharm and Cansino vaccines from China, and the Sputnik V vaccine from Russia, in its Gotong Royong vaccination drive.
Source:
 The Kimia Farma personnel suspected of reusing old rapid test samples on travelers at North Sumatra's Kualanamu International Airport, along with samples of their wrongdoing, at a police press conference in the North Sumatra provincial capital Medan on Thursday (29/4/2021)
The Kimia Farma personnel suspected of reusing old rapid test samples on travelers at North Sumatra's Kualanamu International Airport, along with samples of their wrongdoing, at a police press conference in the North Sumatra provincial capital Medan on Thursday (29/4/2021)